Scandion Oncology

Chemotherapy overcoming drug resistance
Summary edit edit source
Scandion Oncology is a Danish biotechnology company that specialises in developing drugs to reverse chemotherapy resistance. Its lead asset, SCO-101, is being investigated as an add-on therapy to existing treatments in metastatic colorectal cancer (mCRC) and unresectable pancreatic cancer (PC). Management’s clinical programme is spearheaded by the Phase II CORIST study in FOLFIRI-resistant mCRC, from which Edison Investment Research expects crucial proof-of-concept results in Q322. The company is also pursuing the Phase Ib PANTAX trial in PC. Assuming proof-of-concept is met, the positioning of SCO-101 in higher lines of mCRC treatment will be essential to maximise value, in Edison Investment Research's view. Edison values Scandion Oncology at SEK586.5m or SEK18.3 per share.
| Year end | Revenue
(DKKm) |
PBT*
(DKKm) |
EPS*
(DKK) |
DPS
(DKK) |
P/E
(x) |
Yield
(%) |
|---|---|---|---|---|---|---|
| 12/20 | 1.0 | (21.5) | (0.53) | 0.0 | N/A | N/A |
| 12/21 | 0.8 | (57.2) | (1.61) | 0.0 | N/A | N/A |
| 12/22e | 0.8 | (60.1) | (1.70) | 0.0 | N/A | N/A |
| 12/23e | 0.8 | (114.7) | (3.40) | 0.0 | N/A | N/A |
Proof-of-concept incoming edit edit source
Scandion Oncology with its lead asset SCO-101 is targeting indications where there exists significant unmet medical need and resistance to chemotherapy is common. Part 2 of Scandion Oncology’s CORIST study in mCRC will report top-line data in mid-2022. The study is investigating the effect of SCO-101 in tumours that have become resistant to treatment with FOLFIRI, representing last-line therapy. Positive results here would represent the first clinical proof-of-concept for SCO-101 and represent a significant near-term catalyst, in Edison Investment Research's view.
Further studies will require fresh capital edit edit source
Edison Investment Research's estimates see the company funded to complete the ongoing CORIST and PANTAX clinical trials. However, Edison Investment Research expects the company will need a fresh capital injection in 2023 to conduct larger studies. CORIST is an open-label study and Edison Investment Research expects potential Big Pharma partners would likely prefer to review randomised data prior to striking a deal. Edison Investment Research expects the company will require an additional Phase II (or Phase II/III) trial beyond CORIST to obtain a licensing deal targeting the major US and European markets. Edison Investment Research notes that as of 1 June 2022, the company is undergoing a rights issue (for c DKK54m), which is 80% committed.
edit edit source
Edison Investment Research's valuation is based on a risk-adjusted net present value calculation for SCO-101 in last-line mCRC (SEK8.5 per share) and unresectable metastatic PC (SEK5.8 per share) plus a cash position of DKK88.0m at end-March 2022. Edison Investment Research foresees considerable value uplift potential should the company succeed in repositioning SCO-101 to earlier lines of mCRC therapy. At the company’s current burn rate (DKK17.9m in Q122) Edison Investment Research estimates a cash runway into Q123.
Investment summary edit edit source
Company description: Binary inflection point approaches edit edit source
Founded in 2017 and based in Copenhagen, Denmark, Scandion Oncology is focused on developing novel solutions to address chemotherapy resistance in oncology. Management’s present development programme revolves around one asset, SCO-101, which is in trials investigating its use in the treatment of mCRC (Phase II) and PC (Phase Ib). SCO-101 is a first-in-class chemotherapy add-on that is designed to inhibit two well-documented mechanisms of chemotherapy resistance. Scandion’s hypothesis asserts that if these mechanisms are blocked, a chemotherapeutic response will be restored, and patients will see improved survival. There are currently no drugs targeting chemotherapy resistance on the market.
Edison Investment Research's investment thesis rests on crucial proof-of-concept data from part 2 of the CORIST Phase II trial in last-line mCRC, following initial dose finding results from part 1 in 2021. Edison Investment Research expects Scandion demonstrating a clinically meaningful reversal of resistance to FOLFIRI in these patients could result in a material re-rating of the stock. Proof-of-concept data could, in turn, increase confidence in SCO-101’s second indication, PC, for which it is currently in a Phase Ib dose finding trial (PANTAX). Assuming positive results from CORIST part 2, Edison Investment Research believes the company will need to generate randomised data in mCRC in further trials to maximise the value of SCO-101 to potential partners/licensors, targeting the core US and European markets. To fund this, Edison Investment Research expects management would need to raise additional capital and/or pursue non-dilutive smaller, regional licensing deals. In the long term, Edison Investment Research sees the prospects of SCO-101 in other indications and combinations as potentially significant; data from the PANTAX study will be important in corroborating this view.
edit edit source
Edison Investment Research values Scandion Oncology at SEK586.5m or SEK18.3 per share. Edison Investment Research's valuation is based on a risk-adjusted net present value calculation for SCO-101 in wild-type RAS, last-line mCRC (SEK8.5 per share) and unresectable metastatic PC (SEK5.8 per share). Additionally, Edison Investment Research includes a cash position of DKK88.0m at end-March 2022. Edison Investment Research uses a discount rate of 12.5% and assume a licensing deal for SCO-101 will be realised in 2024, assuming positive results from the current Phase II trial.
Financials: Funded into Q123, rights issue pending edit edit source
With a cash burn of DKK17.9m in Q122, a cash position of DKK88.0m and estimated spending in FY22 roughly in line with FY21 (which had a cash burn rate of DKK50.1m), Edison Investment Research estimates a cash runway to Q123. Edison Investment Research anticipates management will raise DKK115m in 2023 to fund randomised trials before a partner is found in 2024 and Edison Investment Research models the financing as illustrative debt. Indeed, as of June 2022, management is undertaking a rights issue which (in the event of full subscription) will provide the company with net proceeds of DKK53m (SEK76.7m: SEK93.7m less SEK17m in transaction costs). Assuming full subscription, management estimates that this cash injection will fund the company into 2024. However, Edison Investment Research notes this may vary according to trial timelines.
At the time of writing, the rights issue is approximately 80% guaranteed. Management intends to use the proceeds from this issue to expand the clinical development of SCO-101 into earlier lines of therapy and mutant-RAS patients in mCRC. Edison Investment Research anticipates a global licensing deal for SCO-101 in 2024 after randomised data is collected in further clinical trials during 2022/23. Edison Investment Research anticipates the company will require roughly an additional DKK200m through 2026 to 2027 to be self-sustaining, excluding potential partnerships. Edison Investment Research expects the company to conduct Phase II in 2023 and have assumed management will need to engage global big pharma partners to largely fund the bulk of its Phase III programmes in 2024.
Sensitivities: Pureplay biotech risks edit edit source
Scandion Oncology is subject to the regular risks associated with drug research and development. As a pureplay biotech, the company will be affected by development delays or failures, regulatory risks, competitor successes, partnering setbacks and financing risks. The largest development sensitivities relate to the company’s sole clinical asset, SCO-101. The most prominent near-term risk would be failure to demonstrate clinical proof-of-concept in the Phase II CORIST trial. Scandion may need to raise capital beyond Edison Investment Research's forecasts. While Edison Investment Research's model accounts for financing(s) as long-term debt, the company may need to issue equity instead, at pricing that may not be favourable for current shareholders and could lead to significant dilution.
Scandion Oncology: The cancer resistance company edit edit source
Two shots on goal for SCO-101 edit edit source
Scandion Oncology’s clinical development strategy for SCO-101 revolves around two clinical studies: CORIST (NCT04247256) and PANTAX (NCT04652206) (Exhibit 1). Of these, the Phase II CORIST trial is the most advanced and aims to demonstrate proof-of-concept for SOC-101 in patients with mCRC in combination with FOLFIRI (folinic acid + fluorouracil + irinotecan). Colorectal cancer represents the third leading cause of cancer-related deaths in the United States. Colorectal cancer has an estimated incidence rate of 0.0039% in the United States. In 2020 915,880 people worldwide died from the disease and it is expected that a further 52,580 people in the United States will die from the disease in 2022. At diagnosis, c 20% of patients will have mCRC and five-year survival of patients with mCRC is only 20%, representing a clear unmet medical need.
Exhibit 1: SCO-101 development strategy[2]
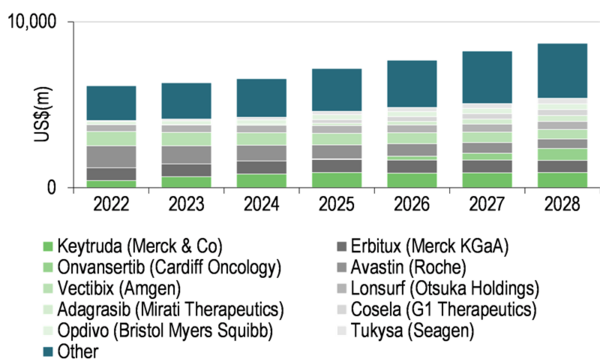
The market for CRC treatment is estimated to reach $8.7bn by 2028 (EvaluatePharma) and, since Avastin (bevacizumab, Roche) went off-patent in 2019, has become more fragmented (see Exhibit 2). This market represents an attractive opportunity for SCO-101, in Edison Investment Research's view, as Edison believes Scandion could potentially establish market share for its product candidates more easily compared to indications that are dominated by one or two therapies. Additionally, many treatments for CRC (including mCRC) are already off-patent, for example irinotecan and 5-fluorouracil (the active ingredients of FOLFIRI). Edison Investment Research believes this will motivate Scandion to pursue premium pricing for SCO-101 as it would generally be used in combination with relatively inexpensive drugs.
Exhibit 2: Estimated CRC market to 2028[3]
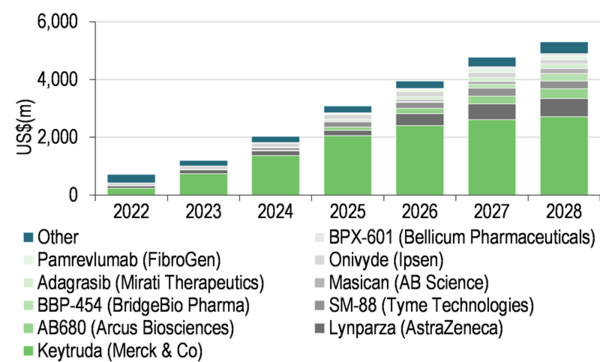
Exhibit 3: Estimated PC market to 2028[3]
The second trial, PANTAX, is a Phase Ib study investigating the safety and tolerability of SCO-101 in patients with unresectable or metastatic PC as an add-on to treatment with nab-paclitaxel and gemcitabine. PC accounts for only 3% of all cancers but 7% of cancer-related deaths, due to the disease’s invasive and often incurable nature. Accordingly, the five-year survival rate for patients with PC is only 11%, dropping to 3% for patients who are diagnosed after the disease has metastasised. The PC treatment market is estimated to reach $5.3bn by 2028 (source: EvaluatePharma). However, Merck’s Keytruda (pembrolizumab) is expected to dominate the market in this period (consensus worldwide sales in 2028 of $2.7bn). Edison Investment Research notes that, as in CRC, many standard PC treatments are off-patent (eg nab-paclitaxel) and if SCO-101 can restore clinical response to these (eg nab-paclitaxel), Edison Investment Research sees an sizable opportunity for Scandion Oncology in this market.
Partnership deal will be key for development edit edit source
The company awaits an important proof-of-concept readout from part 2 of the Phase II CORIST clinical trial in mCRC, which is expected in mid-2022. Success in CORIST will be key in establishing proof-of-concept and advancing it to subsequent trials. Edison Investment Research notes that CORIST is an open-label study and Edison Investment Research believes potential global big pharma partners will prefer randomised data before considering in-licensing, meaning further clinical studies will likely be necessary, in its view, to maximise the value of SCO-101. Assuming positive data from CORIST part 2, Edison Investment Research anticipates management will raise additional capital to fund randomised trial (likely Phase II) thus building the data package needed to engage global big pharma partners.
IP position encouraging edit edit source
Around 20 years ago, SCO-101 (then known as endovion) was a clinical compound under investigation for the treatment of sickle cell anaemia and was owned by Danish biotechnology company NeuroSearch. The drug was discontinued after Phase I trials. Subsequently, Scandion Oncology was formed in 2017 after initial investigations indicated the compound as a potential add-on therapy to overcome cancer resistance. Scandion now possesses a large intellectual property estate on SCO-101, which it is continuously expanding. Importantly, the company has market protection for the treatment of cancers with SCO-101 in combination with other anti-cancer agents until 2037 in Europe (EP3458052B and EP3622953B) and 2038 in the US (US11,103,481). Edison Investment Research sees this as a positive position for Scandion as it will (assuming the potential launch of SCO-101 in CRC in 2026) allow for over 10 years of market protection in Europe and the United States.
Combatting chemotherapy resistance edit edit source
The rapidly dividing nature of cancer cells means chemotherapeutic resistance is commonly observed in oncology. The mechanisms are varied and resistance is a widely recognised problem when treating cancer, so much so that the company asserts 90% of cancer deaths are caused by chemotherapy resistance. The development of drug-resistant cancer will almost inevitably lead to more aggressive tumours and poor prognosis. Resistance can be classified as intrinsic or acquired based on the point at which resistance develops. Intrinsically resistant cancers have developed chemotherapeutic resistance before treatment has been administered, whereas acquired resistance is built up over time and is driven by exposure to a specific chemotherapeutic regimen.
Drug-resistance mechanisms are commonly reported edit edit source
Cancer drug resistance is a complicated process that can occur through a variety of underlying biological mechanisms. Exhibit 4 presents a selection of common drug resistance types seen in oncology.
| Resistance type | Underlying mechanism (example) | Effect | Common drugs affected |
|---|---|---|---|
| Multi-drug resistance | Up-regulation of ATP Binding Cassette membrane proteins (ABC). | Increased removal of chemotherapy from tumour cells. | Doxorubicin, vinblastine, paclitaxel, irinotecan |
| Inhibition of cell death | Down-regulation of pro-cell death proteins (eg p53). Up-regulation of anti-cell death proteins (eg Bcl-2/xl). | Increased tumour cell survival, even in the presence of toxic agents. | Cisplatin, doxorubicin |
| Altered drug metabolism | Altered activity of UGT enzymes. Increased activity of cytochrome P450 enzymes. | Increased metabolism of active agents reduces effectiveness of chemotherapy. | Docetaxel, gemcitabine, cytarabine |
| Enhanced DNA repair | Increased activity of nucleotide excision repair system and homologous recombination repair mechanisms. | Repair of damaged DNA causes increase in tumour cell survival. | Cisplatin, doxorubicin |
In addition to these mechanisms, chemotherapeutic resistance can arise due to alterations to a drug’s target, gene amplifications and epigenetic alterations. Modern approaches to overcoming resistance have focused on developing new, targeted drugs and immunotherapies (for example PD-(L)1 and CTLA4 monoclonal antibodies). However, drug resistance in chemotherapy continues to be recognised as an area of unmet medical need.
SCO-101 has a dual mechanism of action edit edit source
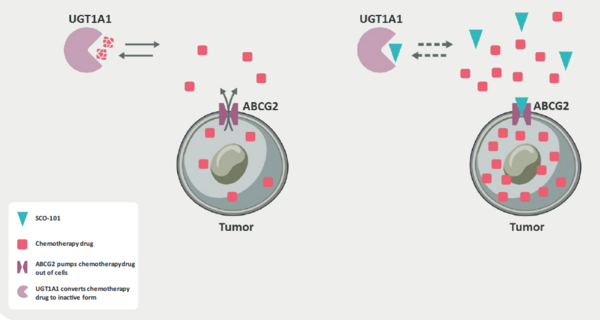
With its lead asset SCO-101, Scandion Oncology is targeting two mechanisms of drug resistance in oncology, multi-drug resistance and altered drug metabolism. Scandion claims that SCO-101 is a first-in-class chemosensitiser, a class of compounds that when used in combination with traditional chemotherapy agents may re-sensitise resistant cancer cells to treatment. Specifically, SCO-101 is a potent inhibitor of the ATP Binding Cassette G2 (ABCG2) efflux pump and UTG1A1 enzyme.
As a member of the ABC superfamily of transmembrane efflux pumps, ABCG2 (also known as the breast cancer resistance protein, BCRP) is important in the development of chemotherapeutic resistance. In normal tissues, ABCG2 is involved in a range of functions including cell protection (in the brain and placenta), homeostasis, nutrient absorption, and hormone regulation. However, when overexpressed in tumour cells, ABCG2 mediates drug resistance through the increased removal of cytotoxic agents from the cell interior (Exhibit 5). Furthermore, many classes of chemotherapeutics are proven substrates for ABCG2. Examples include topoisomerase I and II inhibitors (irinotecan and doxorubicin, respectively), DNA intercalators, nucleoside analogues (clofarabine), thymidylate synthase inhibitors (5-fluorouracil), EGFR inhibitors (erlotinib) and PARP inhibitors (rucaparib), among others. In Edison Investment Research's view, the prevalence of ABCG2-mediated drug resistance highlights the opportunity for SCO-101 in combination with chemotherapies other than FOLFIRI.
Exhibit 5: SCO-101 dual mechanism of action[5]
Additionally, as stated above, SCO-101 inhibits the enzyme UDP-glucuronosyltransferase 1A1 (UGT1A1) to block the metabolism of chemotherapy drugs and increase systemic levels. The UGT family of enzymes work by modifying a chemotherapy drug with glucuronic acid, therefore making it more water soluble and easier to eliminate from the body. There is a body of clinical evidence that supports the role of UGTs in the development of drug resistance in multiple classes of chemotherapy agents, including alkylating agents, targeted therapies, antiangiogenics and hormonal therapies. Altogether, SCO-101’s dual mechanism acts to reverse drug resistance by increasing the level of chemotherapy drug in the body (through UGT1A1 inhibition) while simultaneously decreasing the ABCG2 mediated removal of the drug from the cell.
CORIST: A tale of two parts edit edit source
Scandion Oncology’s Phase II CORIST study is a multi-centre, open-label, dose-escalation study of SCO-101 in combination with FOLFIRI that will enrol up to 50 patients with mCRC. CORIST is targeting last-line mCRC treatment, in which enrolled patients will have failed all prior chemotherapy due to resistance and are in the terminal stages of the disease. Considering the small population of patients at this stage, Edison Investment Research expects the company will look to position SCO-101 in earlier lines of treatment in later studies, provided there are positive results in Phase II.
The CORIST study itself consists of two parts. Part 1, a dose escalation (3+3) study that concluded in H121, demonstrated a maximum tolerated dose (MTD) and a good safety profile for SCO-101 in combination with FOLFIRI. Importantly, part 1 identified wild-type RAS (wtRAS) as a potential biomarker for SCO-101 treatment. In this trial, patients with wtRAS tolerated higher doses of the SCO-101/FOLFIRI combination and stayed on treatment longer. Five out of eight patients identified with wtRAS also showed stable disease for more than eight weeks, compared to progressive disease in all mutant RAS patients. Mutations in RAS are common oncogenic drivers that are regularly screened for during cancer diagnosis. Scandion has used this information to inform CORIST part 2, which is underway and will enrol 25 mCRC patients harbouring wtRAS to investigate the efficacy of the SCO-101/FOLFIRI combination at the MTD. Despite this, Scandion indicated in June 2022 that it plans to also pursue the opportunity with SCO-101 in the mutant-RAS population as this could potentially double the addressable patient population for SCO-101. Edison Investment Research anticipates that it investigate starting an additional mCRC trial in mutant RAS patients.
The primary endpoint for part 2 will be objective response rate (ORR), defined as complete response (CR) and partial response (PR) using RECIST v 1.1. Secondary endpoints include progression free survival (PFS), duration-of-response, overall survival (OS) and further biomarker analysis. Positive results from CORIST part 2 would provide clinical proof-of-concept for SCO101 use in mCRC and will begin to define how Scandion Oncology will position the drug for further development. Top-line results from CORIST are expected in Q2/Q322 (most likely Q322).
Data support FOLFIRI combination edit edit source
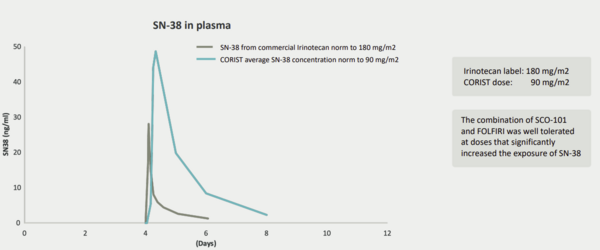
First-line therapy for mCRC relies heavily on 5-fluorouracil (5FU)-based chemotherapy regimens, often supplemented with targeted therapies (should a molecular driver be present) and/or biologic agents (eg vascular-endothelial growth factor A, VEGF, targeting monoclonal antibody, bevacizumab). Later lines of therapy will also generally be formed around modification of 5FU-based regimens. A common treatment in this setting is FOLFIRI, a chemotherapy regimen consisting of leucovorin calcium, 5-fluorouracil and irinotecan. One of the active ingredients of this regimen, irinotecan, is a prodrug of the potent topoisomerase I inhibitor SN-38. Drugs of this class kill tumour cells by inhibiting DNA repair processes. The development of resistance to irinotecan chemotherapy through ABCG2 efflux pump overexpression and UTG1A1 activity is well documented, thus Scandion Oncology has identified FOLFIRI as a potentially attractive combination therapy for SCO-101. Indeed, analysis of CORIST patients treated with SCO-101 and irinotecan in Part I of CORIST has shown dramatically increased plasma levels of SN-38 at a 90mg/m2 dose when compared to SN-38 data from treatment with irinotecan at 180 mg/m2 (based on patients outside the CORIST study, Exhibit 6), although Edison Investment Research cautions that there are limitations to comparing data from different studies.
Exhibit 6: SN-38 plasma concentrations in CORIST patients[6]
While plasma concentrations in the CORIST patients were dramatically higher when compared to patients treated only with irinotecan (using data compiled from separate studies), Edison Investment Research notes that high plasma levels of SN-38 do not necessarily correlate with a clinical effect, especially in ABCG2 driven resistance. However, this data does provide support for the SCO-101/FOLFIRI combination with the lower 90mg/m2 CORIST dosing regimen for irinotecan (compared to 180mg/m2 commercial norm).
Positioning is key to unlocking SCO-101 value in mCRC edit edit source
Should data from the CORIST clinical trial prove positive, Edison Investment Research expects Scandion to begin positioning SCO-101 in earlier lines of therapy. To this end, the company intends to conduct positioning studies in the second line of treatment for mCRC, likely in combination with FOLFIRI ± VEGF and/or EGFR antibodies, before moving to pivotal Phase III trials. Edison Investment Research believes repositioning SCO-101 to the second line in mCRC could add significant value to SCO-101. Edison Investment Research notes, the small patient group size in CORIST part 2 (n=25) and open-label study design will, in its view, likely require Scandion to perform larger, randomised studies to maximise deal value with potential development partners and/or licensors. The company plans to initiate pivotal Phase II/III trials in second-line mCRC patients in 2023, provided data from Part 2 of CORIST are supportive. Furthermore, as stated previously, in June 2022 management announced its intention to use the proceeds from a rights issue to expand SCO-101 clinical development to RAS-mutant patients.
PANTAX: Resistance in pancreatic cancer edit edit source
Scandion Oncology’s second clinical trial, PANTAX, is investigating SCO-101 in the treatment of unresectable or metastatic PC. PANTAX is a Phase Ib, open-label, dose-escalation (3+3) study to establish the safety profile and MTD of SCO-101 in combination with nab-paclitaxel and gemcitabine. The study aims to enrol 18 patients and in addition to primary outcome measurements (safety, MTD) will assess multiple secondary endpoints including, ORR, PFS, OS and pharmacokinetic profile.
There remain significant unmet medical needs in the treatment of PC. If diagnosed as unresectable or metastatic, therapy options are limited for patients. Surgery and radiation therapy may be used to manage symptoms, but chemotherapy is usually the only option to improve survival. Gemcitabine is a common first-line therapy and is often used in combination with other drugs such as erlotinib, cisplatin or nab-paclitaxel. Despite this survival rates remain low. Hence, Edison Investment Research sees PC as a potentially valuable indication for SCO-101 but caveat that it is still in early development. Top-line results from PANTAX are expected in Q2/Q322 (likely Q322). Assuming positive Phase Ib results, the company intends to initiate randomised Phase II trials in PC in 2023.
SCO-101 works in synergy with nab-paclitaxel edit edit source
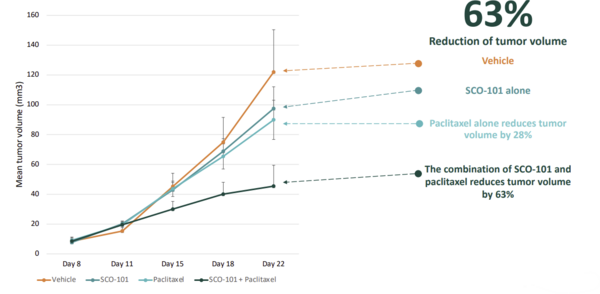
Preclinical data presented by Scandion Oncology at the British Association for Cancer Research (BACR) in September 2021 demonstrate the potential of SCO-101 in combination with nab-paclitaxel (Exhibit 7). In these mouse model data, a paclitaxel/SCO-101 combination demonstrated a 63% reduction in tumour volume compared to control, and paclitaxel alone reduced volume by 28%. In Edison Investment Research's view, these preclinical data support the rationale for the combination of SCO-101 and nab-paclitaxel with gemcitabine in the treatment of PC. Nevertheless, Edison Investment Research notes that interpretation of preclinical results is not a reliable indicator of clinical utility.
Exhibit 7: Preclinical SCO-101 data in combination with paclitaxel[7]
SCO-101 could be first in class in chemotherapy resistance edit edit source
In the past, attention in combatting chemotherapy resistance has focused on repurposing existing therapeutics to serve as add-on treatments to help combat chemotherapy resistance. Specifically, many kinase inhibitors have been investigated as potential combinations to enhance irinotecan response. These include sorafenib, imatinib mesylate, sunitinib, nilotinib and dasatinib. However, to date, none have been approved for use in this setting, due mainly to a lack of efficacy or unfavourable safety profiles.
As a direct comparison to SCO-101 in mCRC, Edison Investment Research draws attention to recent data from the Phase Ib/II trial of onvansertib (Cardiff Oncology) in combination with FOLFIRI and bevacizumab in second-line mCRC patients. These data demonstrate a 35% ORR and 9.4-month mPFS (vs 13% ORR and 5.6-month mPFS historically) in KRAS mutant mCRC patients. While onvansertib works through PLK1 inhibition not ABCG2/UTG1A1 inhibition, Edison Investment Research sees these results as a meaningful comparison for SCO-101. As Scandion intends to pursue both wild-type and mutant RAS mCRC populations, Edison Investment Research believes onvansertib could represent a market competitor in mutant-KRAS mCRC patients (potentially 50%) if both drugs are approved.
There are no approved chemosensitisers on the market. Still, interest in the field means there are drugs in development that may be competition for SCO-101 in future, if approved. Here Edison Investment Research highlights two comparators. Firstly, bemcentinib, an AXL-inhibitor being developed by BerGenBio for the treatment of non-small cell lung cancer, is hypothesised to restore sensitivity to anti-PD-1 immune checkpoint inhibitors. Bemcentinib is estimated for approval in 2023 and to reach worldwide sales of $140m by 2026 (source: EvaluatePharma). Edison Investment Research believes a second, earlier-stage comparator for SCO-101 is ORIC-553 (ORIC Pharmaceuticals), a CD37 inhibitor in Phase Ib trials for the treatment of multiple myeloma.
Preclinical pipeline will build future value edit edit source
In addition to its clinical development programme, Scandion is conducting preclinical studies for the use of SCO-101 in double combination with chemotherapy plus immunoncology. In a setting (oncology) where many higher lines of treatment are dominated by immune checkpoint inhibitors, Edison Investment Research sees this as a natural progression for the company’s development platform. Provided preclinical positive results, Edison Investment Research expects Scandion to work further on developing the SCO-101/immunotherapy combinations. Alongside this, a second oral efflux pump inhibitor, known as SCO-201, is being evaluated in preclinical studies for the treatment of solid tumours.
Sensitivities edit edit source
As a pureplay biotech, Scandion Oncology is subject to all the regular sensitivities associated with drug research and development. The company’s prospects will be affected by development delays or failures, regulator risks, competitor successes, partnering setbacks and financing risks. The drug resistance mechanisms that Scandion Oncology is targeting with SCO-101 are well understood and documented, however this does not ensure success in clinical studies. Failure to demonstrate clinical proof-of-concept for SCO-101 in CORIST represents a significant near-term risk and would likely have significant implications for the company’s pipeline and strategy.
Scandion Oncology does not generate any revenues from its drug discovery and development operations. Consequently, it will require additional capital in future to fund its development objectives. Edison Investment Research models future fund-raisings as illustrative debt, although in June 2022 the company is engaging in an authorised rights issue with current shareholders for 10.7m shares. If exercised in full at the subscription price of SEK8.75, Edison Investment Research expects the issue would result in c SEK76.7m (c DKK53.6m) in net proceeds for Scandion Oncology, considering the company estimated transaction costs would be SEK17m. At the time of writing, the company has received guarantee commitments for 80% of the potential issue proceeds. Subscription of shares will take place between 16 June 2022 up to and including 1 July 2022.
Once the company advances to Phase III trials, expenses are expected to significantly increase. Edison Investment Research forecasts that the company will need to raise c DKK115m (part of which may be covered by funds from the rights issue) in 2022 and will secure a partner in 2024 that will assume R&D funding for Phase III studies and beyond from that point. However, if expenditure trends are above Edison Investment Research's forecasts and/or if a partnership is not secured by its projected timeline, this could lead us to increase Edison's funding expectations.
Valuation edit edit source
Edison Investment Research values Scandion Oncology at SEK586.5m or SEK18.3 per share. Edison Investment Research's valuation is based on a risk-adjusted net present value (rNPV) of SCO-101 and includes cash and cash equivalents of DKK87.97m at end-March 2022. Edison Investment Research's method includes the valuation for SCO-101 in both last-line mCRC (peak sales $295.2m, rNPV of SEK8.5 per share) and metastatic PC (peak sales $685.0m, rNPV of SEK5.8 per share) in both the US and EU5 markets, using a discount rate of 12.5%. Until management confirms its intention to treat mutant-RAS type patients through the initiation of a study in this population, given the data gathered in the CORIST study to date, Edison Investment Research only includes wtRAS patients in its valuation for mCRC. The assumptions used in Edison Investment Research's valuation are presented in Exhibit 8.
| Indication | Comments |
|---|---|
| Last-line mCRC |
|
| mPC |
|
Edison Investment Research's rNPV assumes a licensing deal for SCO-101 in EU5 and the United States is completed in 2024, with Scandion funding randomised Phase II development work until mid-2024, before the partner assumes responsibility for all subsequent R&D and commercialisation expenses. Based on an analysis of recent Phase II and III licensing deals in CRC (Exhibit 9, EvaluatePharma), Edison Investment Research assumes a total deal value of $200m; consisting of an upfront payment of $20m, development milestones of $60m and sales milestones of $120m over the period of patent protection, which Edison Investment Research includes in its rNPV.
| Deal date | Company | Product | Deal partner | Status on deal date | Upfront payment ($m) | Deal value
($m) |
|---|---|---|---|---|---|---|
| 01/06/2021 | Zai Lab | Adagrasib | Mirati Therapeutics | Phase III | 65 | 338 |
| 20/01/2021 | Elpiscience | ES104 | TRIGR Therapeutics | Phase II | 7 | 117 |
| 07/12/2020 | 3D Medicines | Zeltherva | Sellas Life Sciences Group | Phase III | 8 | 202 |
| 25/06/2019 | Ono Pharmaceutical | CPI-613 | Rafael Pharmaceuticals | Phase III | 13 | 163 |
| 03/12/2018 | TRIGR Therapeutics | ES104 | ABL Bio | Phase II | 5 | 595 |
| 14/08/2018 | HLB LifeScience | Aitan | Bukwang Pharmaceutical | Phase III | 9 | 37 |
| 03/04/2018 | Pint Pharma | Nerlynx | Puma Biotechnology | Phase II | 10 | 35 |
| 12/02/2018 | OncXerna Therapeutics | Bavituximab | Avid Bioservices | Phase II | 8 | 103 |
| Average (mean) | 16 | 199 |
Further, given the respective stages of development, Edison Investment Research assumes a success rate of 20% for mCRC and 15% mPC. From these assumptions (Exhibit 10), Edison Investment Research's valuation for Scandion Oncology is composed of 47% mCRC, 32% mPC and 21% cash. Edison notes that if Scandion successfully repositions SCO-101 into second-line mCRC, the relative portion of its valuation assigned to CRC could increase dramatically.
| Product | Indication | Launch | Peak | Peak sales ($m) | Value
(SEKm) |
Probability | rNPV
(SEKm) |
rNPV/share (SEK) |
|---|---|---|---|---|---|---|---|---|
| SCO101 | mCRC | 2026 | 2030 | 295.2 | 1,478.2 | 20% | 274.1 | 8.5 |
| SCO101 | PC | 2029 | 2033 | 685.0 | 1,452.5 | 15% | 185.9 | 5.8 |
| Net cash at 31 March 2021 | 125.7 | 100% | 125.7 | 3.9 | ||||
| Valuation | 3,025.2 | 586.5 | 18.3 | |||||
Financials edit edit source
In FY21, Scandion reported an operating loss of DKK55.4m, a 132% increase year-on-year (FY20 operating loss: DKK23.8m) due mainly to growing clinical costs driven by R&D expenses associated with the CORIST trial. In Q122 the company reported grant funding of DKK0.1m and an operating loss of DKK16.3m, up 65% y-o-y (Q121: DKK9.9m) again due to increasing R&D costs associated with the clinical program. Assuming a similar expense rate as Q122 (DKK16.4m) in future, Edison Investment Research expects an operating loss for FY22 of DKK60.1m, slightly up year-on-year. Edison Investment Research forecasts an R&D expense for FY22 of DKK52.5m (FY21: DKK47.7m) coinciding with the conclusion of CORIST part 2 and PANTAX studies, and keep administrative costs flat year-on-year at DKK8.5m.
Further, Edison Investment Research expects R&D expenses to increase dramatically to DKK107.0m in FY23. Edison Investment Research apportions two-thirds of this figure (DKK75m) to the start of randomised Phase II trials in mCRC based on enrolling 100 patients at a cost of DKK750,000 (c $105,000) per patient. The remaining DKK32m R&D expense in FY23 Edison Investment Research assigns to the commencement of a Phase II trial in PC, based on a similar cost base as CORIST part 2. In total, Edison Investment Research forecasts operating expenses of DKK115m for FY23. In FY24 Edison Investment Research expects an additional six months of R&D expense to be incurred by the company at the same rate as FY23 (leading to DKK53.5m in R&D costs in FY24), until a licensing deal is completed mid-2024, after which Edison Investment Research expects the partner to fund further development of SCO101. These are Edison Investment Research's estimates and may vary with management’s discussions with potential partners. Edison Investment Research notes that its trial timeline forecasts for starting the Phase III programmes (2024) differ slightly from the company’s communicated objectives, where it could potentially initiate pivotal Phase II/III trials in second-line mCRC patients with wtRAS in 2023.
Scandion has communicated it is funded to conclude current clinical work and will not begin new trials until extra funding is sourced. Therefore, at the current burn rate of c DKK17.5m per quarter and with a cash position of DKK87.97m, Edison Investment Research estimates a runway into Q123. Edison Investment Research therefore expects the company to raise extra capital in FY22 to fund further clinical development, which Edison Investment Research has modelled as illustrative debt of DKK115m in its forecast. Edison Investment Research sees this amount as sufficient to fund operations (at a similar cost rate to FY23) to mid-2024, when Edison Investment Research forecasts the company will engage in a global licensing deal. Once the rights issue has completed, Edison Investment Research will adjust its forecasts.
As of June 2022, the company is engaging in an authorised rights issue with current shareholders for 10.7m shares. If exercised in full at the exercise price of SEK8.75 Edison Investment Research expects the issue would result in c SEK76.7m (c DKK53.6m) net in cash for Scandion Oncology (c SEK93.7m less SEK17m in transaction costs). At the time of writing, the company has guaranteed commitments for 80% of the potential rights proceeds. Edison Investment Research expects the issue to be completed by early July 2022, at which point Edison Investment Research will update its forecasts. If the rights issue is fully exercised, Edison Investment Research anticipates that it will extend the company’s cash runway to late-2023 (from Q123) and note this may extend into 2024 depending on trial timing. Longer term, Edison Investment Research anticipates the company will require an additional DKK200m (roughly) through 2026 to 2027 to be fully self-sustaining (on recurring SCO101-related commercial revenue), excluding milestone or upfront payments from potential partnerships.
| Accounts: IFRS, year-end: December 31, DKK’000s | 2020 | 2021 | 2022e | 2023e |
|---|---|---|---|---|
| PROFIT & LOSS | ||||
| Total revenues | 1,003 | 797 | 797 | 797 |
| Cost of sales | 0 | 0 | 0 | 0 |
| Gross profit | 1,003 | 797 | 797 | 797 |
| Total operating expenses | (24,758) | (56,164) | (60,933) | (115,453) |
| Research and development expenses | (21,672) | (47,711) | (52,480) | (107,000) |
| SG&A | (3,086) | (8,453) | (8,453) | (8,453) |
| EBITDA (normalized) | (23,474) | (54,763) | (59,754) | (114,274) |
| Operating income (reported) | (23,755) | (55,367) | (60,136) | (114,656) |
| Operating margin % | N/A | N/A | N/A | N/A |
| Finance income/(expense) | 2,233 | (1,846) | 0 | 0 |
| Exceptionals and adjustments | 0 | 0 | 0 | 0 |
| Profit before tax (reported) | (21,522) | (57,213) | (60,136) | (114,656) |
| Profit before tax (normalised) | (21,522) | (57,213) | (60,136) | (114,656) |
| Income tax expense (includes exceptionals) | 4,384 | 5,508 | 5,508 | 5,508 |
| Net income (reported) | (17,138) | (51,705) | (54,628) | (109,148) |
| Net income (normalised) | (17,138) | (51,705) | (54,628) | (109,148) |
| Basic average number of shares, m | 32.1 | 32.1 | 32.1 | 32.1 |
| Basic EPS (DKK) | (0.53) | (1.61) | (1.70) | (3.40) |
| Adjusted EPS (DKK) | (0.53) | (1.61) | (1.70) | (3.40) |
| Dividend per share (DKK) | 0.00 | 0.00 | 0.00 | 0.00 |
| BALANCE SHEET | ||||
| Tangible assets | 136 | 386 | 338 | 306 |
| Intangible assets | 0 | 0 | 0 | 0 |
| Right-of-use assets | 312 | 1,215 | 1,215 | 1,215 |
| Other non-current assets | 148 | 314 | 314 | 314 |
| Total non-current assets | 596 | 1,915 | 1,867 | 1,835 |
| Cash and equivalents | 5,814 | 105,710 | 164,645 | 54,044 |
| Current tax receivables | 4,384 | 5,500 | 5,500 | 5,500 |
| Trade and other receivables | 1,414 | 2,018 | 2,018 | 2,018 |
| Other current assets | 174,513 | 1,076 | 1,076 | 1,076 |
| Total current assets | 186,125 | 114,304 | 173,239 | 62,638 |
| Non-current loans and borrowings | 8 | 0 | 0 | 0 |
| Non-current lease liabilities | 0 | 500 | 500 | 500 |
| Other non-current liabilities | 504 | 84 | 84 | 84 |
| Total non-current liabilities | 512 | 584 | 584 | 584 |
| Accounts payable | 26,064 | 4,580 | 4,580 | 4,580 |
| Illustrative debt | 0 | 0 | 115,000 | 0 |
| Current lease obligations | 316 | 723 | 723 | 723 |
| Other current liabilities | 3,962 | 5,791 | 5,791 | 5,791 |
| Total current liabilities | 30,342 | 11,094 | 126,094 | 11,094 |
| Equity attributable to company | 155,867 | 104,541 | 48,428 | 52,795 |
| CASH FLOW STATEMENT | ||||
| Operating income | (23,755) | (55,367) | (60,136) | (114,656) |
| Depreciation and amortisation | 281 | 604 | 382 | 382 |
| Share based payments | 0 | 0 | 0 | 0 |
| Other adjustments | 4,223 | 2,899 | 4,023 | 4,023 |
| Movements in working capital | 2,024 | 2,066 | 0 | 0 |
| Cash from operations (CFO) | (17,227) | (49,798) | (55,731) | (110,251) |
| Capex | (46) | (318) | (334) | (351) |
| Acquisitions & disposals net | 0 | (167) | 0 | 0 |
| Other investing activities | 0 | 0 | 0 | 0 |
| Cash used in investing activities (CFIA) | (46) | (485) | (334) | (351) |
| Capital changes | 7,892 | 150,690 | 0 | 0 |
| Debt Changes | 0 | 0 | 115,000 | 0 |
| Other financing activities | (226) | (511) | 0 | 0 |
| Cash from financing activities (CFF) | 7,666 | 150,179 | 115,000 | 0 |
| Cash and equivalents at beginning of period | 15,421 | 5,814 | 105,710 | 164,645 |
| Increase/(decrease) in cash and equivalents | (9,607) | 99,896 | 58,935 | (110,601) |
| Effect of FX on cash and equivalents | 0 | 0 | 0 | 0 |
| Cash and equivalents at end of period | 5,814 | 105,710 | 164,645 | 54,044 |
| Net (debt)/cash | 5,806 | 105,710 | 164,645 | 54,044 |
Management team edit edit source
President and CEO: Bo Rode Hansen
Bo Rode Hansen brings more than 20 years of international biotech and pharma business experience. Before joining Scandion Oncology, he most recently served as president and CEO at Genevant Sciences Inc. Prior to Genevant Sciences, he served as global head of Roche RNA Therapeutics and general manager of Roche Innovation Center Copenhagen. Before Roche, Bo served as executive, VP and head of drug discovery & alliances at Santaris Pharma, until the company was acquired by Roche.
CFO: Johnny Stilou
Johnny Stilou has held numerous executive positions as chief financial officer within the biotech and pharmaceutical industry, most recently as CFO at Amgen Research Copenhagen and Nuevolution AB (acquired by Amgen). Prior to Nuevolution, he served as CFO at Veloxis Pharmaceuticals until the company was acquired by Asahi Kasei.
COO and head of R&D operations: Maj Hedtjarn
Maj Hedtjärn brings more than 15 years of experience in drug discovery and development, program management and pharma alliance management from the biotech and pharmaceutical industry (Roche, Santaris Pharma and Lundbeck), where she has held numerous leadership positions within R&D. Prior to joining Scandion Oncology, she most recently served as VP and head of drug discovery, RNA Therapeutics Research at Roche Innovation Center Copenhagen.
CMO: Alfredo Zurlo
Alfredo is a senior pharma and biotech medical executive with more than 20 years’ experience in clinical development and medical affairs. After leaving his academic roles at the Italian University in 1999, Alfredo worked as medical advisor at the EORTC Data Center in Brussels. In 2003, he joined Roche in Basel as medical director in charge of the launch of bevacizumab (Avastin) in Europe and rest of the world for the colorectal cancer indication. Having held several senior positions at the Basel headquarters and the French affiliate over the course of the years, Alfredo left Roche and started consulting in 2011 for several pharma and biotech clients, until he became the CMO of Mologen in 2013, and later of Glycotope in 2016.
| Principal shareholders | % |
|---|---|
| SANIONA AB | 10.81 |
| Stenvang Jan | 4.33 |
| Brunner Nils | 3.52 |
| AVANZA PENSION | 3.38 |
| NORDNET PENSIONSFORSAKRING | 2.47 |
| Tang-Jespersen Christian | 1.63 |
| Goeran Ofsen | 1.17 |
References edit edit source
- ↑ Note: *PBT and EPS are normalised, excluding amortisation of acquired intangibles, exceptional items and share-based payments.
- ↑ Source: Scandion Oncology interim report Q122.
- ↑ 3.0 3.1 3.2 Source: EvaluatePharma, Edison Investment Research.
- ↑ 4.0 4.1 4.2 Source: Edison Investment Research.
- ↑ Source: Scandion Oncology corporate presentation.
- ↑ Source: Scandion Oncology CMD update.
- ↑ Source: Scandion Oncology BACR presentation September 2021.
- ↑ Source: Scandion Oncology company accounts, Edison Investment Research.